British pharmaceutical company AstraZeneca corona vaccine tested successfully, available for 1 billion doses
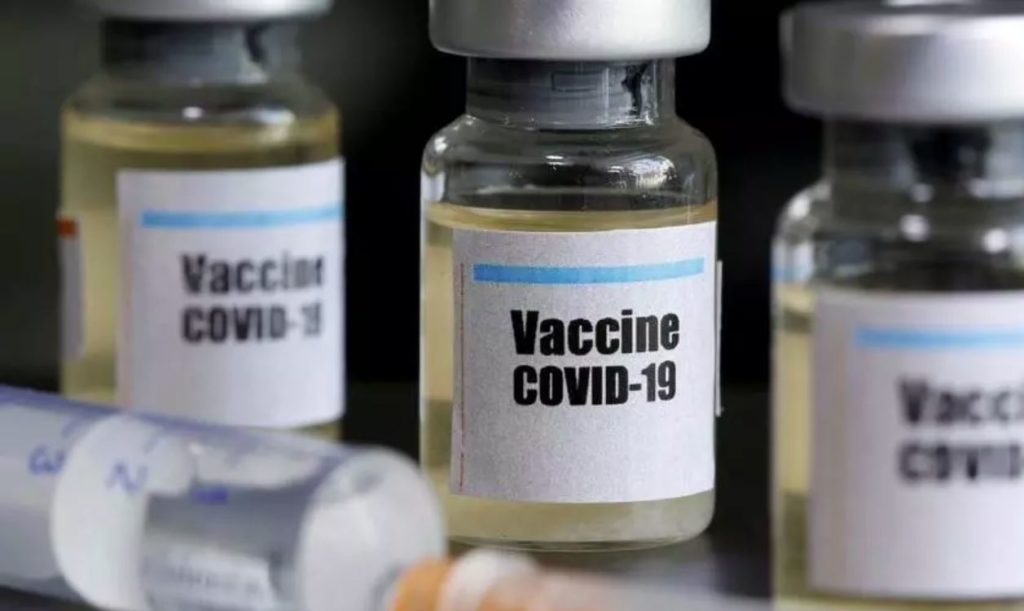
AstraZeneca, a British pharmaceutical company, said on May 21 that the first clinical trials of the new corona vaccine they developed should bring results soon. It is estimated that if the final test is successful, 1 billion doses will be delivered this and next year.
AstraZeneca said the vaccine, developed in collaboration with the University of Oxford in the UK, has signed its first supply contract and will deliver at least 400 million doses.
AstraZeneca acknowledges that this vaccine may ultimately prove ineffective, but if the initial test results are optimistic, it may be carried out in several countries in later trials.
Currently, only a few pneumonia vaccines being developed in Wuhan have been tested in humans, which means they are safe and effective, but most of them also fail at this stage.
Large international pharmaceutical companies, including Pfizer, Johnson & Johnson and Sanofi, have also invested in research and development of pneumonia vaccines in Wuhan, and are currently at various stages.
In addition, US pharmaceutical company Inovio Pharmaceuticals said yesterday that they developed an experimental pneumonia vaccine in Wuhan that produces protective antibodies and immune system responses when tested in mice and guinea pigs.
Moderna, an American biotechnology company, also published optimistic test data for this week’s candidate vaccine, showing that a small group of healthy volunteers produced protective antibodies.
AstraZeneca also said in a statement on the 21st that they received funding of more than $ 1 billion. USA from the United States Office of Biomedical Research and Development (BARDA), which will be used for research and development, production, and supply of this Candidate Vaccine.
Animals recovered from infection with the new coronavirus
American researchers have announced that two studies on monkeys may provide some initial scientific evidence that recovering from infection with a new coronavirus can develop immunity to avoid reinfection. This optimistic trait indicates that a new crown vaccine is being developed that may be successful. , These studies have been reviewed. The study does not prove that people will develop immunity, and how long it can last, but such results are still encouraging.
Reuters said good news! The monkeys who recovered after being infected with the new coronavirus developed immunity, and the vaccine was successful.
According to the report, although scientists believe that antibodies produced by the body in response to a new corona have a protective effect, there is little rigorous scientific evidence to support this view. In a recent study, researchers infected nine monkeys with a new corona. After the monkeys recovered, the research team again exposed them to the virus. They found that the monkeys did not have any reproductive diseases.
Dr. Dan Barush of the Center for Virology and Vaccine Research, a subsidiary of the Beth Israel Deaconess Medical Center at Harvard Medical School, said the results of the study show that they “actually produce natural immunity to avoid reinfection.” The results of their research were published in the journal Science. Baruch said: “This is very interesting news.”
In a second study, Barush and colleagues tested six vaccine prototypes on 25 monkeys to see if the resulting antibodies are protective.
Also Read:
Guo Mingji: Apple iPhone 12 will cancel the random included AirPods
Good news! Monkeys recovered from infection with new coronavirus develop immunity
YouTube app now reminds you to go to sleep






