Good news! New coronavirus oral vaccine has been developed
China facing many problems such as downgrade of the market due to the new crown virus. Many people also died due to this virus and thousand of people are effective from this. China try to work to develop the antidote of the coronavirus and they are closer to the success
Obviously Good News, Some laboratories have announced that they have developed an oral vaccine for the new crown virus.
Jinyun News reports that Tianjin University’s School of Life Sciences professor Jinhai Hai team has announced that they have successfully developed a new oral virus for the coronavirus. In which food-grade safe sclerosis secretes as a carrier. As a target for generating protein antibodies. Professor Jinhai, who has taken oral doses of the new crown vaccine sample 4 times with no side effects.
At the moment, the scientific research team is looking for partners. Hoping to get the vaccine to clinical as soon as possible and to play a role in the prevention and control of epidemic diseases.
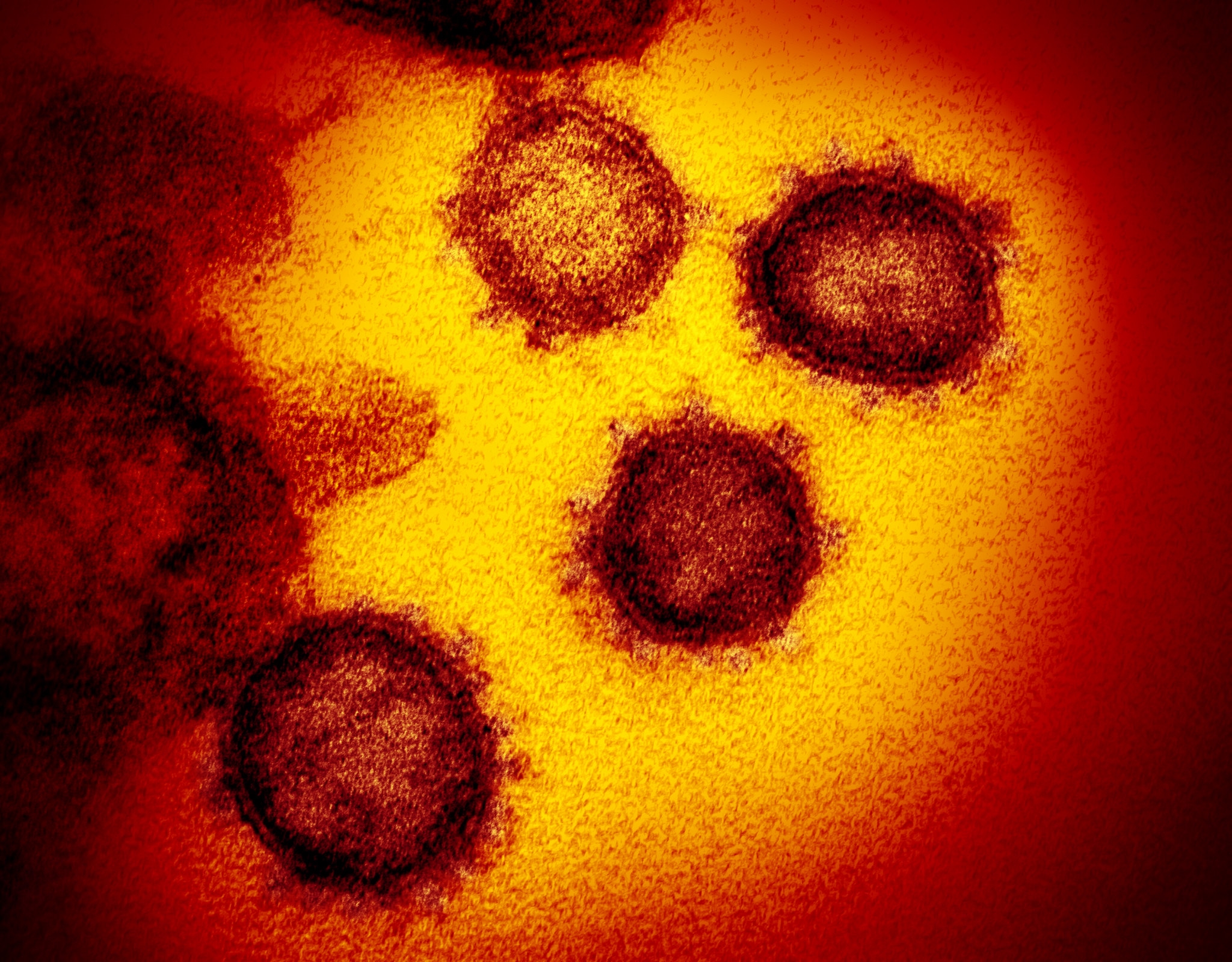
The new Coronavirus S protein (spike protein) can bind to the host’s viral receptors. It is an important protein that determines the invasion of virus-sensitive cells and is a key target for new coronavirus vaccines and drug development for prevention or treatment.
According to Professor Huang, in addition to the foreign genes of the virus. The expression factor of the vaccine is all fragments of the SCRC’s own gene and does not include resistant genes. The new coronavirus can be used as a therapeutic treatment for viral infections, through the high expression of protective antigens, and it can also act as a preventative immune system by activating a preventive immune vaccine mechanism. Its safety, immunization, cost savings and other benefits are exempt. And it is especially suitable for emergency and prevention and control of chronic diseases without traditional vaccines.
Vaccines are a tough and long process from development to market. Currently, the team is reviewing animal toxic, immune efficacy and pilot preparation activities. It is hoped that eligible businesses and units will cooperate to complete the vaccine testing process and accelerate the development of the vaccine. Clinical validation and popularity will help prevent and control this epidemic.
Also, Read
All Info: Sony has announced a new mid-range smartphone – Sony Xperia 1 II + Specification
China approved two new coronavirus antibody kits: Zhong Nanshan announces good news!






